chromatography
-
Why Choose ICP-OES Coupled with Chromatography
Inductively Coupled Plasma Optical Emission Spectroscopy (ICP-OES) is widely recognized as one of the most powerful techniques for determining elemental concentrations in a variety of sample matrices. However, when combined with chromatography, this pairing enhances the analytical capabilities of ICP-OES, enabling more detailed analysis of complex samples. This article explores the benefits, principles, and applications of coupling ICP-OES with chromatography.
Understanding ICP-OES
ICP-OES is an atomic emission spectroscopy technique used to detect and quantify metals and some non-metals in liquid samples. In ICP-OES, a sample is introduced into a plasma where it is atomized, and the emitted light from the excited atoms is measured. The intensity of light at specific wavelengths corresponds to the concentration of the elements present in the sample. ICP-OES is particularly valued for its high sensitivity, wide dynamic range, and ability to handle a broad range of elements simultaneously.
Chromatography: An Overview
Chromatography, on the other hand, is a technique used to separate compounds within a mixture based on their chemical properties. Several types of chromatography exist, such as gas chromatography (GC), liquid chromatography (LC), and ion chromatography (IC). These methods are primarily used for separating complex mixtures into individual components.
Chromatography’s key advantage is its ability to separate, identify, and quantify specific substances in a sample, making it an ideal tool when dealing with complex samples that may contain multiple interfering substances. By separating the components of a mixture before analysis, chromatography helps ensure that only the target analytes are measured, improving the accuracy and reliability of the results.
The Benefits of Coupling ICP-OES with Chromatography
The combination of ICP-OES with chromatography provides a powerful tool for analyzing complex samples, offering several advantages:
1. Improved Sensitivity and Resolution
Chromatography is excellent at separating components within a complex sample. By combining it with ICP-OES, the analysis can focus specifically on the elements of interest that have been separated from other substances, leading to improved sensitivity and resolution. This combination helps avoid matrix effects or spectral interferences that may be present when analyzing complex samples directly with ICP-OES alone.
2. Increased Analytical Versatility
Coupling ICP-OES with chromatography broadens the scope of analysis. While ICP-OES is adept at detecting metals and other elements, chromatography can separate complex organic compounds, ions, or molecules. When coupled, this combination can be used for a variety of applications, including the analysis of environmental, biological, pharmaceutical, and industrial samples.
3. Enhanced Quantification
ICP-OES provides precise quantitative analysis of elemental concentrations, and when chromatography is used to separate components, it allows for more accurate quantification of individual analytes in mixtures. For example, by separating a sample into its constituent components first (via chromatography) and then detecting the elements within those components (using ICP-OES), a much higher level of accuracy can be achieved.
4. Reduced Interference
Chromatographic separation can eliminate interferences caused by matrix components that could affect the accuracy of ICP-OES results. These interferences may come from high concentrations of coexisting elements or complex sample matrices that could overlap with the analyte’s emission wavelengths. Chromatography minimizes this problem by isolating the elements of interest, enabling clearer and more precise emission measurements in ICP-OES.
Common Chromatographic Techniques Used with ICP-OES
There are several chromatography techniques commonly paired with ICP-OES, each tailored to specific types of analysis:
1. Ion Chromatography (IC)
Ion chromatography is widely used for the separation of ions in aqueous samples. When coupled with ICP-OES, it allows for the precise separation and detection of metal ions, making it ideal for environmental analysis, water quality testing, and pharmaceutical applications. The separation of ions can reduce interference, allowing for more accurate measurements of trace metal concentrations.
2. High-Performance Liquid Chromatography (HPLC)
High-performance liquid chromatography is one of the most commonly used chromatographic techniques in combination with ICP-OES. It is particularly effective for separating organic compounds or metal-organic complexes. When coupled with ICP-OES, HPLC can be used to analyze a variety of samples, including food, pharmaceuticals, and environmental samples, allowing for simultaneous detection of both organic and inorganic components in a sample.

3. Gas Chromatography (GC)
Although less common, GC can also be coupled with ICP-OES for the analysis of volatile compounds. This pairing is particularly useful for detecting organometallic compounds, such as methylmercury or organotin compounds, where the GC separates the organic components, and the ICP-OES analyzes the metal content.

Applications of ICP-OES Coupled with Chromatography
The combination of ICP-OES and chromatography has proven to be a versatile and effective approach in a wide range of industries. Here are some of the most common applications:
1. Environmental Analysis
In environmental monitoring, ICP-OES coupled with chromatography allows for the analysis of metals in water, soil, and air samples. Chromatography is used to separate complex environmental matrices, while ICP-OES offers the sensitive detection of trace metals like lead, mercury, cadmium, and arsenic. This combination is instrumental in ensuring compliance with environmental regulations and standards.
2. Pharmaceutical and Food Analysis
In the pharmaceutical and food industries, the combination of ICP-OES and chromatography helps in the analysis of metal contamination in products. For example, HPLC can separate different organic compounds in a food sample, and ICP-OES can then measure the levels of heavy metals that may be present in those compounds. This is essential for ensuring the safety and quality of pharmaceutical and food products.
3. Clinical and Biological Analysis
ICP-OES coupled with chromatography is also widely used in clinical and biological research. For example, in the analysis of blood or urine samples, chromatography can separate different proteins or metabolites, and ICP-OES can detect trace elements like calcium, magnesium, or potassium. This is valuable for diagnosing nutrient deficiencies or monitoring drug levels in the bloodstream.
4. Industrial Applications
In industrial settings, ICP-OES coupled with chromatography can be used to monitor metal concentrations in various manufacturing processes. It helps in ensuring product quality, monitoring environmental discharges, and controlling raw material quality. It is commonly used in the analysis of metals in oils, lubricants, and other industrial fluids.
Conclusion
Coupling ICP-OES with chromatography enhances the capabilities of both techniques, making them more powerful tools for the analysis of complex samples. This combination offers improved sensitivity, better separation, reduced interference, and the ability to analyze both organic and inorganic components in a single analysis. Whether for environmental monitoring, pharmaceutical quality control, or industrial analysis, the synergy of ICP-OES and chromatography continues to prove invaluable in achieving more accurate, reliable, and comprehensive results.
PR -
How Advancements in Sample Preparation are Revolutionizing GC-MS Analysis
Sample preparation is a critical step in gas chromatography-mass spectrometry (GC-MS) analysis, significantly impacting the accuracy, sensitivity, and reliability of results. Over the years, significant innovations have emerged to address the challenges associated with sample preparation, enhancing the capabilities of GC-MS analysis across various industries.

Challenges in Sample Preparation
1.Matrix Interference: Complex sample matrices can introduce contaminants that interfere with analyte detection.
2.Analyte Stability: Some analytes may degrade or transform during sample handling and storage.
3.Low Analyte Concentrations: Trace-level analysis can be challenging due to low analyte concentrations.
4.Sample Complexity: Complex matrices with numerous components can hinder chromatographic separation.
5.Method Reproducibility: Ensuring consistent results requires standardized procedures and quality control measures.
Recent Advancements
1.Solid-Phase Microextraction (SPME): Innovative SPME fibers and techniques have improved extraction efficiency and selectivity.
2.Microextraction Techniques: Miniaturization and automation have enhanced the speed and accuracy of microextraction methods.
3.QuEChERS Method: Advanced sorbents and extraction solvents have improved analyte recovery and reduced solvent consumption.
4.Automated Sample Preparation Systems: Integrated systems streamline workflows and reduce human error.
Conclusion
The advancements in sample preparation techniques have significantly improved the capabilities of GC-MS analysis. By addressing the challenges associated with sample preparation, researchers and analysts can achieve higher levels of sensitivity, selectivity, and reproducibility in their analytical workflows.
-
What is Gas Chromatography: Outstanding Benefits and General Procedure
Gas chromatography, a powerful analytical technique, employs gas as a mobile phase within a chromatographic column. This core component of gas chromatography significantly influences the overall performance of the technique. Chromatographic columns come in two main types: packed columns and capillary columns.

Packed columns are filled with solid adsorbents or carriers containing fixed liquids. They are versatile, capable of separating compounds with high boiling points like nitrogen, oxygen, hydrogen, and argon. On the other hand, capillary columns offer high separation efficiency by coating the inner wall with a stationary phase liquid. This phase can be solid or liquid, each with distinct adsorption capacities for various components. Examples of weak adsorbents include sucrose, starch, inulin, talc, and potassium carbonate.
Gas chromatography boasts several outstanding advantages:
High Separation Efficiency: It can effectively separate complex mixtures of compounds with closely matched physical and chemical properties. This enables both qualitative and quantitative analyses, sometimes resolving tens or even hundreds of components simultaneously in a single analysis.
High Sensitivity: Gas chromatography can detect impurities at the ppm or even ppb level. Only minute gas or liquid samples, often less than 1 mL, are required for analysis.
Rapid Analysis: With the aid of computers, precise results can be obtained in seconds, making it a swift analytical method.
Wide Range of Applications: Gas chromatography is versatile, capable of analyzing gaseous, volatile liquid, and solid samples. It finds extensive use in organic analysis, covering approximately 20% of organic compounds, and can even analyze some inorganic substances following appropriate transformations.
The General Procedure for Gas Chromatography:
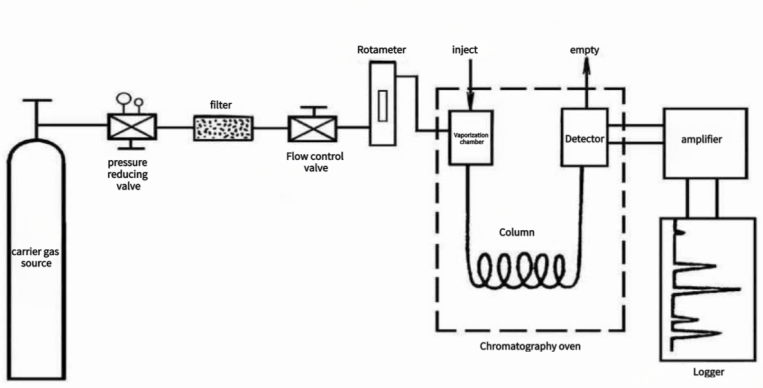
The sequence of peak times serves as the basis for qualitative analysis, while peak area or peak height is used for quantitative analysis.
Three Components of Industrial Gas Chromatography:
Industrial gas chromatography, compared to its laboratory counterpart, incorporates a sampling system, utilizes column-cutting technology, and employs complete automation for program control and information processing.
1. Sampling System:
The sampling system handles sample collection and pretreatment, serving as the interface between the production process and industrial gas chromatography. Initial sample pretreatment steps, such as decompression, water removal, and dust removal, occur external to the chromatography system.
Internally, the gas chromatography system features functionalities such as pressure regulation, flow path switching, flow monitoring, atmospheric balance, and calibration with standard gas or liquid. Design considerations encompass corrosion resistance of system components, leak prevention, explosion protection, reduction of transfer lag time, and control of contamination from evacuation.
2. Carrier Gas Flow System:
The carrier gas flow system comprises a source of carrier gas, purifiers, pressure and flow stabilization, and adjustment devices. Typically, high-pressure gas stored in cylinders serves as the carrier gas source, with a pressure-reducing valve reducing it to 0.1-0.5 MPa. High purity and stability are prerequisites for the carrier gas, necessitating the use of materials like silica gel, molecular sieve, and activated carbon to adsorb moisture and hydrocarbon compounds.
Carrier gases such as hydrogen, nitrogen, and argon may be employed. Maintaining constant gas flow with minimal variation (<1%) is crucial, necessitating the inclusion of flow meters, regulating valves, and pressure-stabilizing valves in the gas path.
3. Sampling Device:
Sampling involves the quantitative introduction of a gas, liquid, or transformed solid sample into the chromatographic column for separation. Consistency in injection quantity, injection duration, and sample vaporization rate significantly impacts the repeatability and accuracy of quantitative results.
(1). Gasification Chamber:
The gasification chamber features a metal block exterior with a controllable operating temperature ranging from 50-500°C. Temperatures exceeding 250°C utilize inner glass tube structures to prevent unwanted catalytic effects. The gasification chamber's primary function is to ensure instantaneous vaporization of liquid samples. It requires preheating of the carrier gas before entry, while the silicone rubber pad should remain cool to prevent unintended chemical reactions.
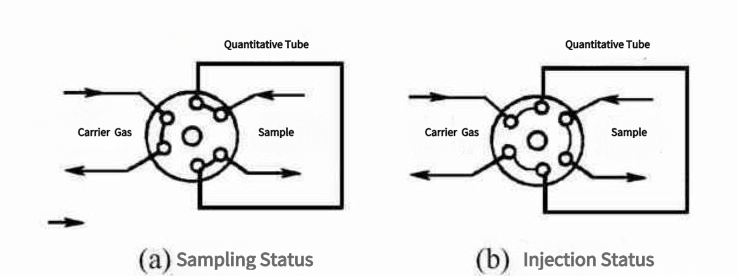
(2). Injection Valve:
Sampling valves must exhibit excellent airtightness, minimal dead volume, reliability, durability, and rapid switching times. In specific applications, resistance to corrosion and operation within defined temperature ranges may also be necessary.
-
Why Select Gas Chromatography (GC) Used in a Wide Range of Industies
Gas chromatography (GC) is a widely used analytical technique for separating and analyzing volatile compounds in a mixture. Different types of detectors and chromatographic columns are employed based on the specific components and properties of the sample being analyzed. Here's a brief summary of each application mentioned:

Gas chromatography analysis of natural gas constants: Uses a thermal conductivity detector to analyze components like O2, N2, CH4, CO2, C2H6, C3H8, i-C4, n-C4, i-C5, n-C5, and others in urban gas.
Analysis of artificial gas: Utilizes a thermal conductivity detector, double-valve multi-column system, and automatic or manual sample injection for determining main components in artificial gas.
Gas chromatography analysis of LPG: Employs thermal conductivity detector, packed column system, and valve automatic or manual switching for analyzing C2-C4 and total C5 hydrocarbons in LPG produced by refineries.
Gas chromatography analysis of refinery gas: Selects thermal conductivity and hydrogen flame ionization detectors, packed column, and capillary column for analyzing various components in refinery gas.
Gas chromatography analysis of benzene and toluene in vehicle and aviation gasoline: Uses thermal conductivity detector or hydrogen flame ionization detector, double columns connected in series, and a backflushing system for qualitative and quantitative analysis of benzene and toluene in gasoline.
Gas chromatography analysis of some alcohols and ethers in gasoline: Utilizes a hydrogen flame ionization detector, multi-column system, and backflushing to analyze alcohols and ethers in gasoline, especially suitable for automotive and aviation gasoline containing ethanol.
Gas chromatographic analysis of hygienic standards for distilled wine and prepared wine: Employs a hydrogen flame ionization detector and specific columns for analyzing methanol and fusel oil in liquor.
Food Alcohol: Uses a PEG-20M capillary column, FID detector, and standard internal method to detect methanol, fusel oil, and other trace components in high-quality edible alcohol.
Analysis of aldehydes, alcohols, and esters in liquor: Utilizes a hydrogen flame ionization detector and specific columns for analyzing components in Luzhou-flavor liquor and Qing-flavor liquor.
Detection of residual solvents in vegetable oils: Employs a headspace gas chromatography system with a hydrogen flame ionization detector and packed column coated with 5% DEGS fixed solution for determining residual No. 6 solvent in leaching oil.
Indoor air detection and analysis: Uses a hydrogen flame ionization detector, thermal desorption sampler, packed column, or capillary column for detecting volatile organic compounds (VOCs) in indoor air.
Gas chromatographic analysis of transformer oil cracking products: Employs hydrogen flame ionization detector and thermal conductivity detector with Ni catalytic converter for automatic analysis of transformer oil cracking products.
Gas Chromatography Analysis of food additives and pesticide residues in food: Utilizes different detectors and chromatographic columns for the detection of various compounds in food samples.
Testing and analysis of tobacco and tobacco products: Employs TCD, FID, and specific chromatographic columns for determining moisture and nicotine content, as well as pesticide residues in tobacco and tobacco products.
Other applications: Includes various analysis methods for ethanol content in blood, residual solvents in medicines, odor analysis in food, volatile aromatic hydrocarbons in wastewater, and volatile organic compounds in drinking water.
These applications demonstrate the versatility and importance of gas chromatography in a wide range of industries, including energy, environmental monitoring, food safety, and pharmaceuticals. Different combinations of detectors, columns, and sample preparation techniques allow for tailored analysis of specific compounds in different sample matrices.
-
What are Common Problems of Ion Chromatography and How to Solve the Problems
Although there are various kinds of ion chromatography on the market, its structure mainly includes five parts: pumping system, sampling system, chromatographic separation column, detector, and data processing. We pay attention to the maintenance of the instrument during its use and operation, to master the ion chromatography analysis technology. Now take the ion chromatograph as an example to introduce the main points, common problems, and solutions of ion chromatography in the use and operation of the equipment.

Two Operation Points of Ion Chromatography
1. Use environmental conditions
Relative humidity: ﹤85%,
Working environment temperature: 15-30℃,
Power supply voltage: 220±10%
It should be equipped with a smooth and flat workbench.
2. Preparations before commissioning
(1) Deionized water or ultrapure water with a conductivity lower than 1μs/cm is required.
(2) The pure water and eluent used should be degassed, which can be degassed by ultrasonic degassing, vacuum pump, water boiling degassing, or online degassing device.
(3) There must be a reliable regulated power supply and grounding wire.
Four Common Problems and Solutions of Ion Chromatography
1. Common faults of conductivity detectors
A common failure of conductivity detectors is the contamination of the detection cell.
Cause of failure: The contaminants mainly come from samples that have not been properly pretreated, such as high concentration and complex sample matrix.
Treatment method:
(1) Wash the conductivity cell with 3mol/L HNO3 solution, and then wash the conductivity cell with deionized water until the pH value is neutral.
(2) Use 0.001mol/L KCl solution to correct the conductivity cell, so that the conductivity value is displayed as 147μS.
2. Analyze the common faults of the pump
Fault phenomenon: The noise of the baseline increases and the shape of the chromatographic peaks becomes worse.
Solution:
The common fault of the analysis pump is the generation of air bubbles and liquid leakage in the pump. Provide sufficient eluent for the analysis pump, and apply a certain pressure (usually less than 35kPa) to the eluent. For solutions that are prone to gas generation, vacuum degassing can be used first, and then inert gas is used for online degassing. If the pump leaks, the pump sealing ring can be replaced.
3. What should I do if the system pressure increases?
Reason: The pressure increase is generally caused by the blockage of the instrument components.
Solution:
When the system pressure is found to increase, it should start from the detector end of the flow path and check one by one to find the specific unit that caused the pressure increase.
For example, when the online filter is blocked, replace the filter element directly. Check whether the peek head in the pipeline is too tight, otherwise, the pressure will increase.
4. Common faults and troubleshooting of ion chromatography suppressor
The suppressor plays a pivotal role in ion chromatography. The working performance of the ion chromatography suppressor has a great influence on the analysis results. The most common failures of suppressors are leakage, which reduces peak area and increases background conductance.
(1) The peak area is reduced
Reason:
The main reasons for the reduction of the peak area are dehydration of the micro membrane, leakage of the suppressor, poor solution flow path, and contamination of the micro membrane.
Solution:
Use a syringe to inject a little 0.2mol/L sulfuric acid solution into the anion suppressor in the opposite direction of the eluent flow path. At the same time, inject a little pure water into the inlet of the regeneration solution, and place the suppressor for more than half an hour. Contaminated metal ions in the suppressor can be cleaned with sodium oxalate.
(2) High background conductance
Reason:
In the process of chemical suppression type conductance detection and analysis, if the background conductance is high, it means that there is a certain problem in the suppressor part. Most are caused by improper operation
Solution:
When the failed suppressor is in use, the background conductance will continue to rise. At this time, a new suppressor should be replaced.
(3) Leakage
Reason:
The main cause of suppressor leakage is insufficient hydration of the micro membrane within the suppressor.
Solution
The ion chromatography suppressor that has not been used for a long time should be allowed to swell with water before use. In addition, it is necessary to ensure that the outlet of the regeneration liquid is smooth. Thus, when the back pressure is large, it will also cause the suppressor to leak. In addition, if the suppressor is improperly stored, the micro membrane in the suppressor shrinks and ruptures, and liquid leakage will also occur.
